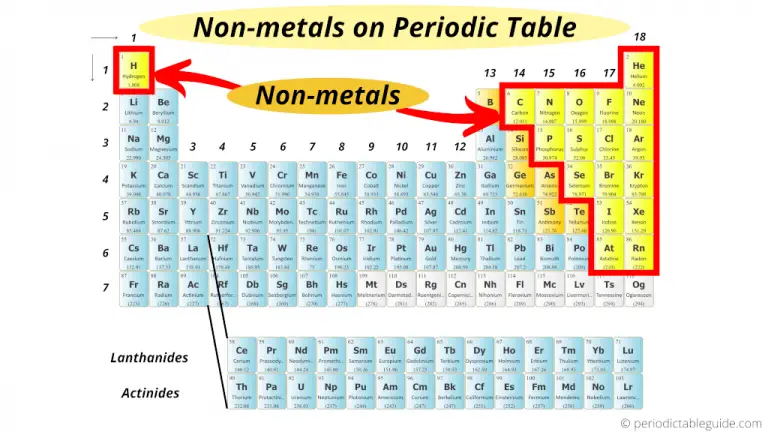
Poor conductors of heat and electricity.Malleable - can be beaten into thin sheets.Good electrical conductors and heat conductors.Nonmetals except the inert gases readily share electrons. These elements do not have the properties of a metal. Nonmetals include the inert gases hydrogen, oxygen, nitrogen, fluorine, and chlorine liquid bromine and the solid elements carbon, sulfur, phosphorus, and iodine. Gain or share valence electrons easily. The metal elements are represented with the color red, and they can be found on the left-hand side of the periodic table. Metals form the major part of the periodic table.A few things to note: In general, elements on the left of the Periodic Table are metals.

Identifying metals and non-metals on the Periodic Table helps us decide the type of compound and the steps we take to name or write its formula. Usually have 4-8 electrons in their outer shell. Metals and Non-Metals on the Periodic Table.Usually have 1-3 electrons in their outer shell.This general description is adapted from Shipman, et al. On the bases of the observations recorded in Table 3.1, discuss the general physical properties of metals and non-metals in the class. It seems appropriate to describe what is meant by "metal" in general terms. Metals and Nonmetals Metals and NonmetalsĪs shown on the periodic table of the elements below, the majority of the chemical elements in pure form are classified as metals.
